Carolin Körner, Jan-Hannes Schäfer, Bianca M. Escha , Kristian Parey, Stefan Walter, David Teis, Dovile Januliene, Oliver Schmidte, Arne Moellerb, Florian Fröhlich
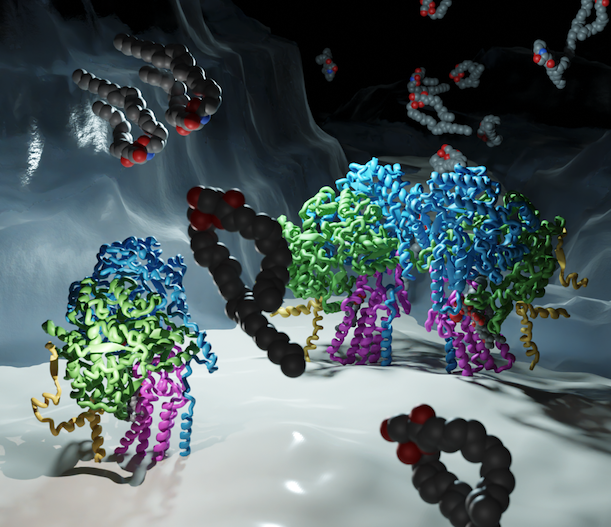
The levels of sphingolipids are crucial determinants of neurodegenerative disorders. Therefore, sphingolipid levels have to be tightly regulated. The Orm protein family and ceramides act as inhibitors in the rate-limiting step of sphingolipid biosynthesis – the condensation of L-serine and palmitoyl-CoA. The two yeast isoforms Orm1 and Orm2 form a complex with the serine palmitoyltransferase (SPT). While the sequences of the Orm proteins are highly similar, however, Orm1 and Orm2 are differentially regulated in yeast cells. The mechanistic details of the differential regulation remain elusive. To elucidate the regulatory mechanism, we determined the cryo-electron microscopy structure of the SPT complex containing Orm2. Through in vitro activity assays and a newly developed plasmid coverage assay combined with targeted lipidomics, we demonstrate that the SPT complex with Orm2 exhibits lower activity in comparison to the complex containing Orm1. This collectively suggests a higher inhibitory potential of Orm2, despite the remarkably similar structures of the Orm1- and Orm2 containing complexes. The high conservation of the SPT from yeast to man implies that the three human ORMDL isoforms could also have different regulatory capacities, which might be essential to understanding their role in sphingolipid-mediated neurodegenerative disorders.