Josephine H. Leung, Lici A. Schurig-Briccio, Mutsuo Yamaguchi, Arne Moeller, Jeffrey A. Speir, Robert B. Gennis, Charles D. Stout
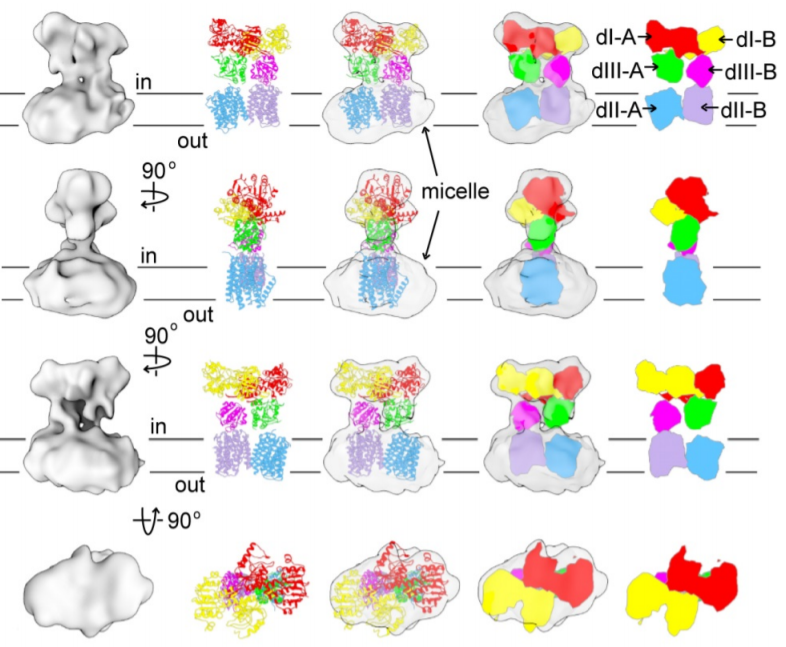
NADPH/NADP+ (the reduced form of NADP+/nicotinamide adenine dinucleotide phosphate) homeostasis is critical for countering oxidative stress in cells. Nicotinamide nucleotide transhydrogenase (TH), a membrane enzyme present in both bacteria and mitochondria, couples the proton motive force to the generation of NADPH. We present the 2.8 Å crystal structure of the transmembrane proton channel domain of TH from Thermus thermophilus and the 6.9 Å crystal structure of the entire enzyme (holo-TH). The membrane domain crystallized as a symmetric dimer, with each protomer containing a putative proton channel. The holo-TH is a highly asymmetric dimer with the NADP(H)–binding domain (dIII) in two different orientations. This unusual arrangement suggests a catalytic mechanism in which the two copies of dIII alternatively function in proton translocation and hydride transfer.
DOI: 10.1126/science.1260451
PMID: 25574024
