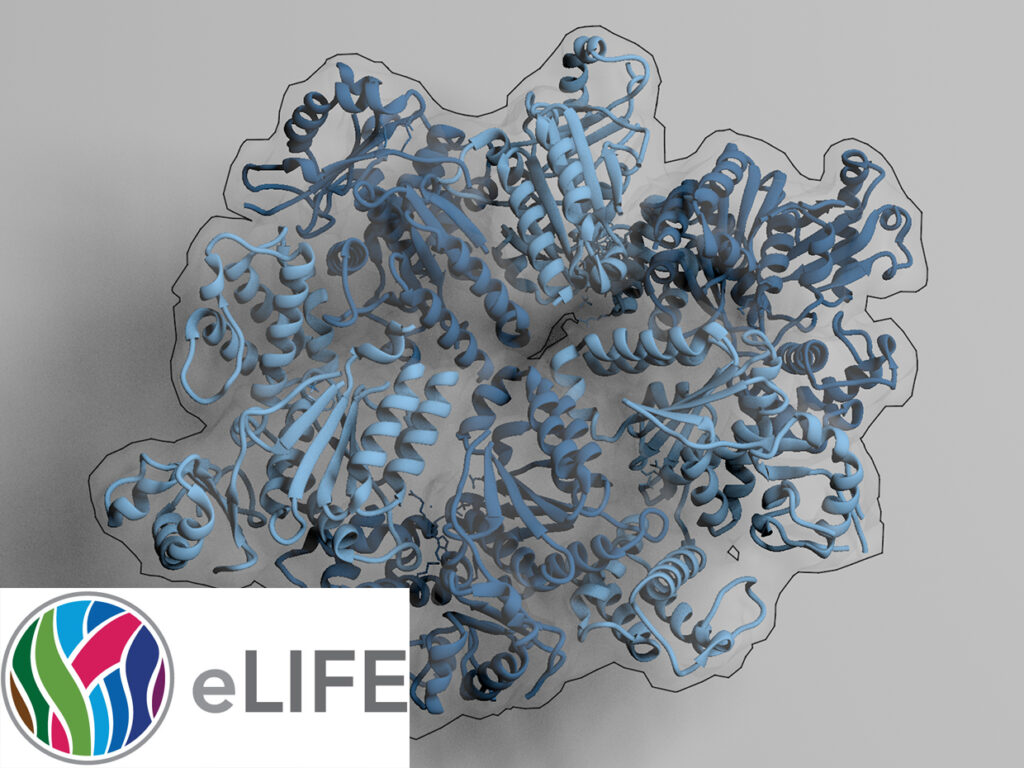
Know your detergents: A case study on detergent background in negative stain electron microscopy
Electron cryo-microscopy (cryo-EM) of purified macromolecular complexes is now providing 3D-structures at near-atomic resolution (Kühlbrandt, 2014). Cryo-EM can tolerate heterogeneous specimens, however, high-resolution efforts demand highly optimized samples.